What is GRHANITE™?
GRHANITE™ is computer software that provides researchers, managers and health professionals with the means to collect data for the purposes of audit, clinical research and health surveillance.
GRHANITE™ is designed to work in any environment where data is being routinely collected and addresses the many complex legal, ethical, organisational and technical barriers that prevent or hinder such vital activities in Australia.
Why is GRHANITE™ needed?
Organisations will not admit to it, but taking shortcuts in acquiring data is commonplace. The Australian public are very vocal in expressing and advocating their rights to privacy and confidentiality and there are many valid historical reasons why this is the case.
Melbourne University and the HIU share the scepticism of the general public and our aim is to do things right once-and-for-all. If shortcuts are taken, it is because doing things properly is difficult and expensive. We have undertaken to do the hard bits and commit the resources so that others don't have to.
The philosophy of GRHANITE™ is to provide one technological solution to healthcare data collection for audit, research and health surveillance that works Australia-wide.
What does GRHANITE™ do that is often missing from other solutions?
GRHANITE™ works with most databases technologies
GRHANITE™ has the built-in capability to source data from almost all modern database technologies. Interfaces to specific GP computer systems have been developed.
Database technologies:
| Text files | ODBC | Oracle | Firebird |
| Spreadsheets | JDBC | SQL Server | 4D |
| OleDB | .NET Provider | Interbase | MS Access |
| Paradox | Visual Objects | SQLite | MySQL |
GP Systems:
| Medical Director V2 | Medical Director V3 | GENIE | Best Practice |
| SHIP | Communicare | MedTech32 | practiX |
| Zedmed |
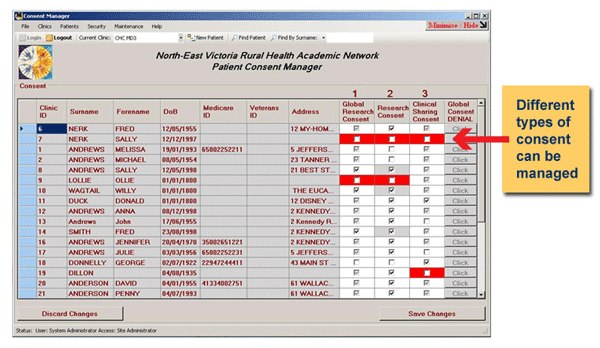
GRHANITE™ manages consent processes
Depending on the consent requirements of a project, GRHANITE™ can help document who has given consent, when and for what purpose. GRHANITE™ will only transmit data where the appropriate permissions are in-place.
- Opt-in consent: If an individual needs to give their permission to be involved in a project, GRHANITE™ can read this consent information if recorded in the clinical database. GRHANITE™ can also provide the clinician with a desktop tool to record this consent if the clinical database is unable to.
- Opt-out consent: If a research project is deemed by ethics committees to be of low risk to the individual, it is commonplace for consent to be managed in an opt-out manner. GRHANITE™ provides mechanisms to implement opt-out consent.
- Waiver of consent: It is sometimes not practical to obtain individual consent. If certain criteria are met, waivers of consent may be granted by ethics committees. Because of the extreme lengths taken by GRHANITE™ to protect identity, this is a common model for GRHANITE™ projects
Back to top
GRHANITE™ ensures study inclusion criteria are applied before data is transmitted
Data to exported can be restricted using any data held in the source database to exactly match study inclusion criteria:
For example:
Extract data for all patients who had two or more consultations in 2008 who were aged 16 - 29 at the time of consultation and who had an HBA1c test undertaken in the year prior to the first consultation
Back to topGRHANITE™ aggregates data at the level required to answer questions
GRHANITE™ is able to analyse data while it is still within the originating clinic organisation. It can aggregate data at any level so that only essential data is exported. This includes removing aggregated data cells where the number of participants is too low to guarantee anonymity.
Back to topGRHANITE™ is one of few systems to provide privacy-protecting record linkage
Australia has no unique identifier that can be used when linking data for research. Because of this, Australian researchers must use identifiers (name, date of birth, sex et-cetera) in any linkage project. The use of such identifiers has many ethical and legal challenges and in some sectors (for example General Practice and Health Departments) such linkages are rarely if ever permitted and are often refused on legal grounds.
Without exporting patient identifiers (name, date of birth, medicare ID, postcode et-cetera), GRHANITE™ is able to generate encrypted keys that can be used to record-link data between 2 or more independent databases.
The encryption used is non-reversible. Because of this, there is no way for the identity of an individual to be ascertained from the information used in the record linkage process.
Because no patient identifiers are involved, it is possible to record link data across jurisdictional boundaries within Australia including General Practice and Health Departments.

Back to top
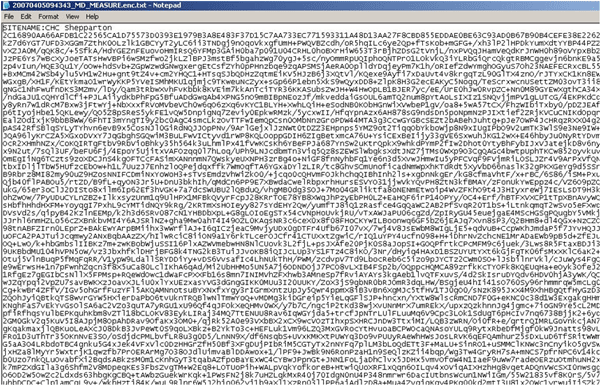
All communications are secured using GRHANITE™
GRHANITE™ uses the same industry and US federal-recommended public/private key encryption algorithms underpinning the banking and eCommerce sectors. This encryption ensures that any data in-transit is secure.
Back to top
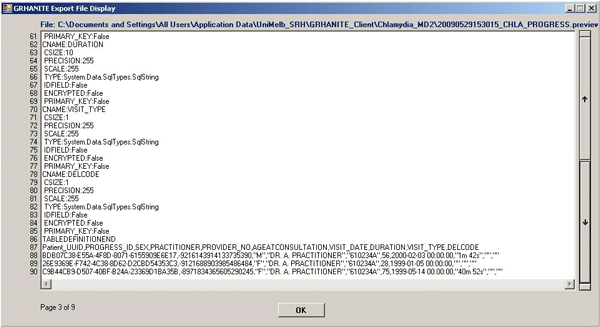
GRHANITE™ has a data previewing facility
GRHANITE™ allows a data custodian to preview data that is due to be transmitted to researchers. This is often a vital part of the process of building trust.
Back to top
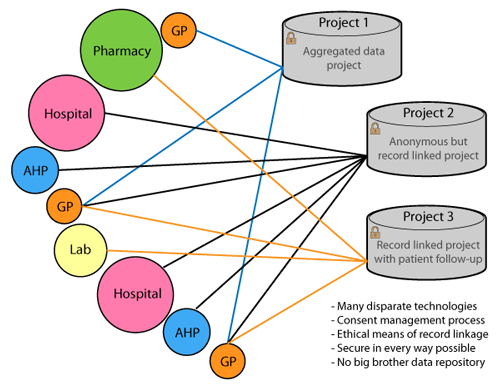
GRHANITE™ ensures data is routed appropriately where and when it is needed
To maintain privacy, it is vital to ensure a researcher is given only enough data to answer a particular research question. Different research projects are kept separate to avoid identity being inferred by the data.
GRHANITE™ sends data to different databases for every project and the recipient computer can be anywhere in the World.
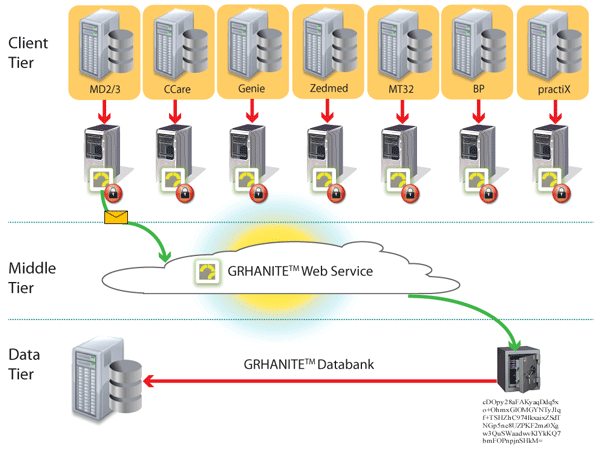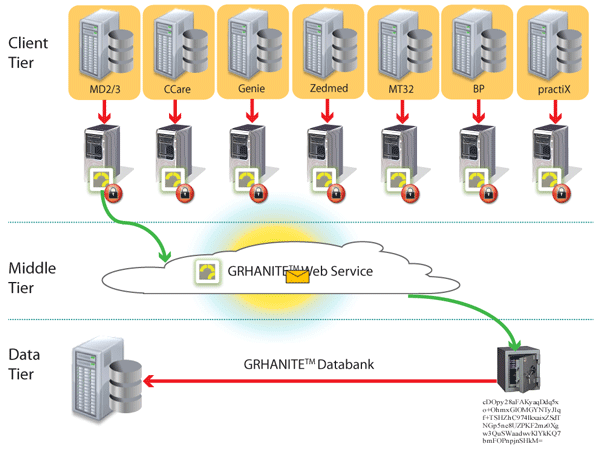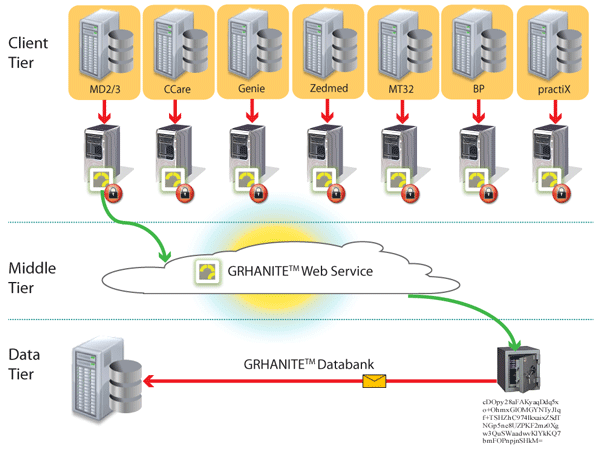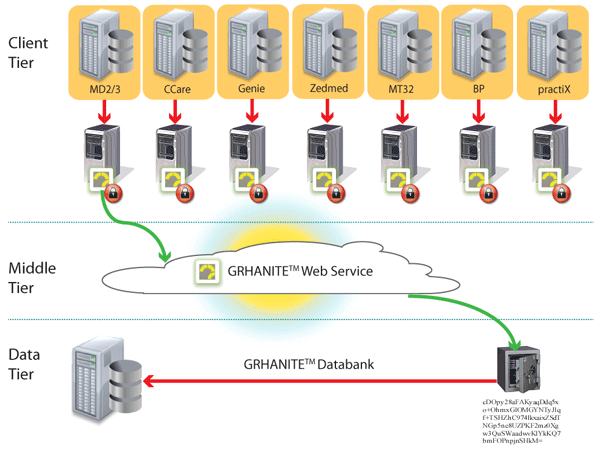
Back to top
GRHANITE™ provides infrastructure tools to support projects over hundreds of participating sites
GRHANITE™ is designed to operate across multiple research projects covering as many as 1000 separate clinical sites. The system has sophisticated tools allowing technical support staff to identify and resolve problems within hours of them occurring.
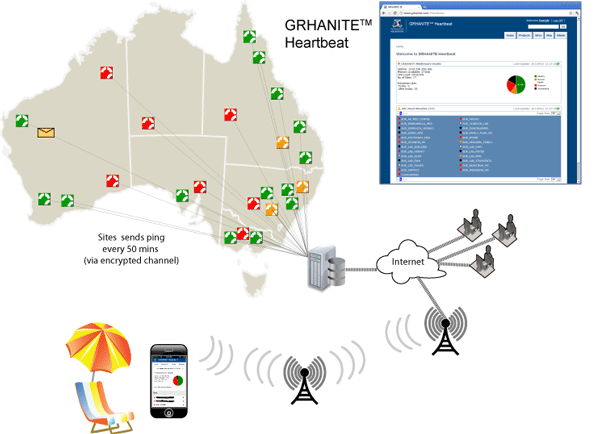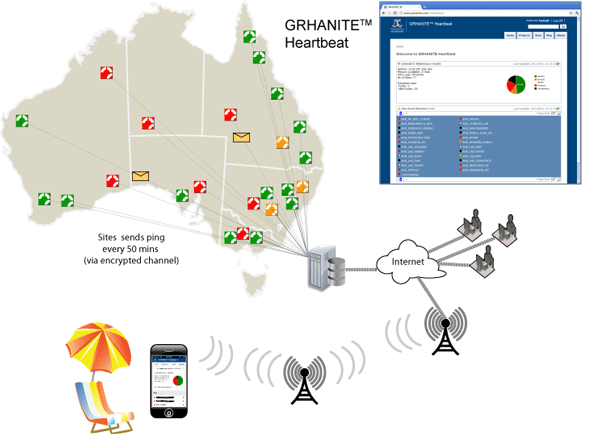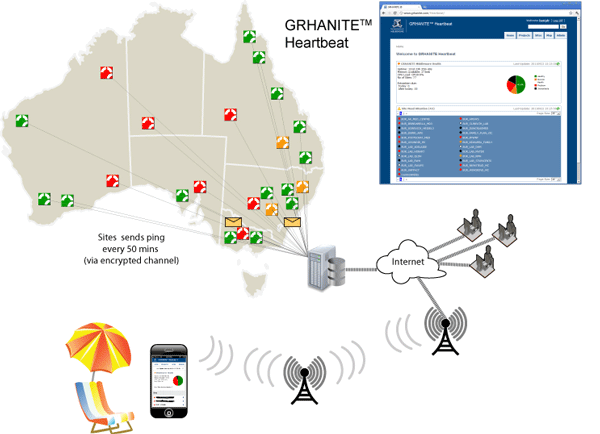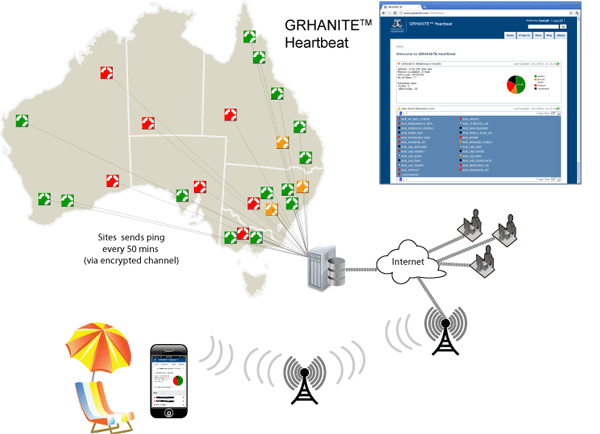
The following are some of the features that allow such a large infrastructure:
- GRHANITE™ has an installer builder that allows installers to be tailor-made for each different project requirement allowing for standardised large-scale deployment.
- Technical support engineers from each research organisation are able to monitor the health of sites participating in their projects. The health is current to within 40 minutes.
- The site health monitoring tool (GRHANITE™ Heartbeat) is web-based and is mobile phone enabled allowing monitoring from anywhere in the World.
- GRHANITE™ data extracts are automated - the system identifies any data extract failures or omissions and highlights these to engineers.
- GRHANITE™ software can be automatically upgraded in a similar way to anti-virus software
- Study protocols can be remotely upgraded - should anything in a study protocol change, these changes can be reflected in the data extraction without visiting the site. This is especially important where sites are geographically distributed.
- Once installed, GRHANITE™ can be used for many projects (install once, use many). Note that data custodian permission and ethics clearance is always required as a prerequisite.